
Research Director: Shoichiro Tsukita
(Professor, Faculty of Medicine, Kyoto University)
Research Term: 1996-2001
Cell biologists often describe the architecture of cells using such words as “beautiful,” meaning the great orderliness and practical placement of the various components for optimized functionality and efficiency. To achieve this the cell must have an axis that defines where eaverything is to be placed as well what is inside and outside the cell. The mystery is how the cell translates the direction imposed by its external environment into an orientation of its internal machinery of its cellular axis. Understanding this mystery was the major objective of this project.
Research Results
The mechanism of action of “unfoldin” which unfolds folded proteins: A new protein was isolated that has the ability to unfold, or denature, a wide variety of enzymes. It has been found that unfoldin is localized in yeast cells in a very polarized manner, suggesting that it plays an important role in regulating the polarity of cells.
Characterization of PCM-1, a component of centriolar satellites: An uncharacterized protein, PCM-1, has been identified as a component of “centriolar satellites,” electron-dense granules around centrosomes, which have not been characterized in molecular terms. The PCM-1-containing centriolar satellites, novel non-membranous organelles, are actively gathered around centrosomes by microtubule-dependent motors. Judging from their organization, they would be important in the function of creating the microtubule organization.
Dynamic behaviors of APC protein and APC-binding protein EB1: GFP fusion protein has been constructed with the protein APC, a tumor suppresser protein. GFP-APC fusion protein has allowed a direct visualization of its peculiar movement along microtubules and drop-like accumulation at their extending tips in moving epithelial cells. Observations have suggested that APC at the cytoplasmic surface of membranes is involved in determining the cell shape and cell polarity.
Targeted disruption of gamma-tubulin gene: Disruption of the gene resulted in early embryonic lethality in mice.
Dynamic features of adherens junctions during Drosophila embryonic epithelial morphogenesis revealed by a D-alpha-catenin-GFP fusion protein: Drosophila catenin was fused to GFP, and then made a transgenic fly expressing GFP-catenin. In the embryonic stage, catenin molecules in the body were directly observed under a confocal microscope. Very interesting video images have been made, showing that adherens junctions are more dynamic than expected. This was the first time to observe catenin behavior at the whole-body level.
Dynamic behaviors of DE-cadherin during Drosophila gasturation: The dynamic aspects of cadeherin-based cell-cell adhesion in morphogenetic events were examined to assess the contribution to morphogenesis. DE- and DN-cadeherin showed complementary expression patterns in the presumptive ectoderm and mesoderm at the mRNA level.
Analysis of PCCD domain which is present in nonchordate classic cadherins: To understand the roles of the PCCD, a series of mutant forms of Drosophila classic cadeherin (DE-cadeherin) was constructed and characterized.
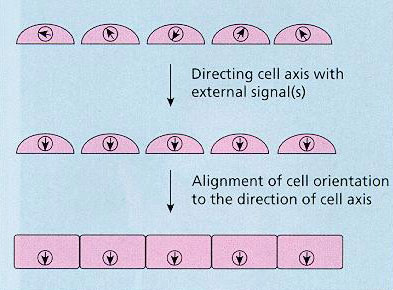
Basic steps of cell organization in tissue
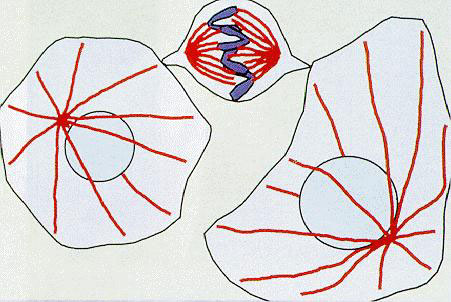
A cell in the process of forming a cell division axis (center) and cells before division (left, right)
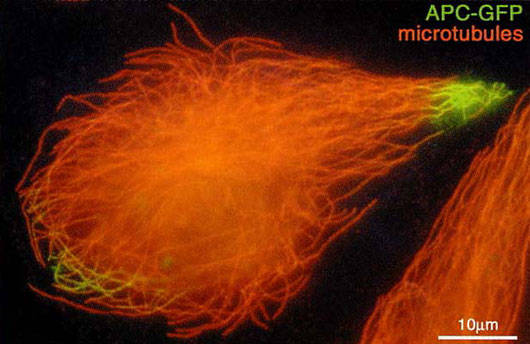
GFP-tagged APC accumulates near the ends of microtubules in the cellular extensions