|
 
 |
|
| Curriculum Vitae |
【 Work Experience 】 |
Present : Designated lecturer, Graduate School of Engineering, Osaka University, Japan
2006 : Designated research associate, OsakaUniversity 21st century COE program “Center for Integrated Cell and Tissue Regulation”, Japan
2005 : Designated postdoctoral fellow, OsakaUniversity 21st century COE program “Center for Integrated Cell and Tissue Regulation”, Japan
|
【 Education 】 |
2005: Graduate School of Engineering, Kyoto University , Japan (Ph.D.)
2000: Graduate School of Pharmaceutical Sciences, Osaka University of Pharmaceutical Sciences, Japan
1998: Osaka University of Pharmaceutical Sciences, Japan |
− Research fields −
laser medicine, cell engineering, biomaterial |
|
| Introduction of the project |
| Stem cells are multipotent cells that replicate as undifferentiated cells and can differentiate to lineages of various tissues, including bone, cartilage, fat, tendon, and muscle. These cells undergo rapid and selective differentiation, characteristics that could be harnessed to provide new therapeutic approaches to restore damaged or diseased tissue. However, the factors that control the differentiation of stem cells are not known. Circadian clock proteins regulate cell cycle, osteogenesis, and adipogenesis in vitro and in vivo. Here we show that blue laser (wave length; 405 nm) irradiation can influence the differentiation of mouse mesenchymal stromal cells (MSCs) into osteoblasts.
|
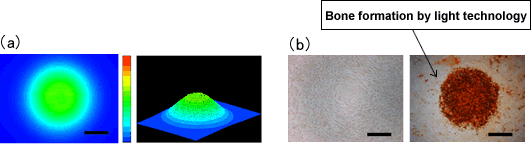 Figure. (a) A beam profile of light used in this research and
Figure. (a) A beam profile of light used in this research and
(b) bone formation of stem cells by light technology. |
| Laser irradiation promoted osteogenesis of MSCs, induced the translocation of circadian rhythm proteins, Cryptochrome, Period, and DEC, from the cytoplasm to the nucleus. I reveal that circadian rhythm proteins are master regulator of the MSCs differentiation regulated by laser irradiation. Now, I report that the MSCs differentiation into not only osteoblasts but chondrocytes and adipocytes can be controlled by laser irradiation. Laser irradiation provides a simple and effective means of controlling the fate of implanted MSCs as a therapeutic strategy. Furthermore, this model system may be useful for exploring the crosstalk between circadian rhythm, bone formation and cell metabolism. |
|
| ▲UP |
|
|
|

