Research Results
Combines with the original bone in a short period after implantation!
Success in the development of sponge-textured artificial bone FY2016
- HOYA Technosurgical Corporation / Junzo Tanaka (Emeritus professor, Tokyo Institute of Technology)
- Development of Creative Technology Seeds, Contract Development
- "Manufacturing technology for biologically substituting organic-inorganic hybrid artificial bone" Developing and implementing company/Representative Researcher (2003-2012)
Strong need for artificial bones with performance higher than autologous bones
Bones support the human body and protect the brain and organs. In cases where a part of these bones is heavily damaged by disease or injury, the bone of the patients (autologous bone) was implanted as a replacement. Since autologous bones can be implanted while maintaining their activities, it is highly functional in bone regeneration, and are associated with significant advantages such as being free of problems related to immunity and infection. However, there is a limit to the amount of autologous bone which can be sampled, and surgeries are required in two locations for the procedure. In addition, there are large burdens on patients, including the frequent problems in the sampling location. For this reason, the idea of artificial bone was conceived.
The artificial bones currently in use are made of ceramic, and have been developed in the 1970's. The percentage of use of artificial bones is around 40% as of 2009. Why are autologous bones still being used? The main reason is that regeneration of bone with artificial bone is not nearly as effective as that of the autologous bone. In Japan, an increasing number of cases require bone grafts (such as bone fracture and bone tumor) with the increased elderly population, and there is a heightened need for artificial bones with performance equal to or superior than the autologous bone. In response to this, an artificial bone based on new ideas were realized by Hoya Corporation, New Ceramics Division (currently Hoya Technosurgical Corporation) based on the results of research by Emeritus Professor Tanaka Junzo and his colleagues, which utilized the Development of Creative Technology Seeds, Contract Development.
Birth of new artificial bone which overcome the problems of operability and insufficient bone regeneration!
There have been previous developments of artificial bones made from various materials, which enable resorption and replacement by bone cells and eventually being regenerated to live bones. An artificial bone using β-type tricalcium phosphate is evaluated highly for the high effect of resorption and replacement; however, there are difficulties of operation during surgery due to its fragility. In addition, there are reports that in some cases only the resorption of the material progressed, resulting in insufficient bone regeneration, or the residual material cause the delay in regenerations. Artificial bone by Emeritus Professor Tanaka and his colleagues was developed to overcome these problems.
Elasticity of the new artificial bone
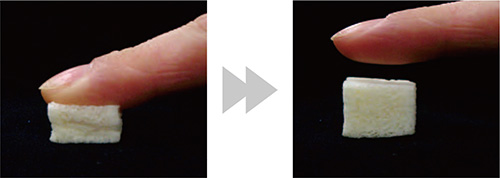
Elasticity never seem before facilitates the handling during surgery
Sponge-like texture with elasticity! Can be cut with a scalpel and integrates quickly with the surrounding bone
The artificial bone developed is created by mixing collagen with thin crystals of hydroxyapatite at a ratio of 4:1, the ratio identical to the live bone, and made into fiber form, resulting in a sponge-like texture. This enables the material to be readily absorbed by the body, and the conjugated fiber with collagen created elasticity that was not observed in conventional artificial bones. As a result, the artificial bone can be processed easily with scalpels and scissors during surgery, and even if the area of bone defect has a complex shape, the artificial bone itself changes shape to fill the area easily and with certainty.
Does this sponge-textured artificial bone function as intended in living bodies? Studies in animals were conducted to confirm the efficacy of the product. A defect at the diameter of 5 mm was created in the shin bone of rabbits, then the artificial bone at 5 mm in diameter and 3 mm in thickness was implanted into this defect, and the condition was observed up to 12 weeks. As a result, the developed artificial bone entered the bone metabolism cycle of the body, and was observed to simultaneously undergo both bone formation around and inside the artificial bone due to osteoblasts and resorption of artificial bone due to osteoclasts, and eventually replaced by autologous bones. The final results showed complete recovery in 60% or more of the cases at 24 weeks after surgery, and the artificial bones in the remaining cases had almost integrated to the surrounding bones. No notable safety problems were observed.
Result of efficacy confirmation study
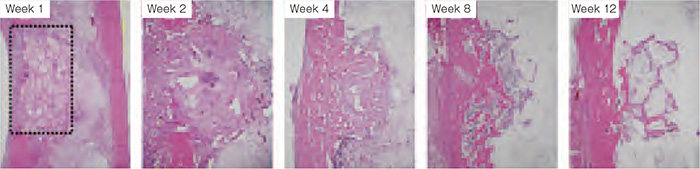
The area shown with broken lines to the left is the area of artificial bone compensation. The artificial bone is resorbed with the passage of time and integrated with the surrounding bones, resulting in replacement by healthy bone by Week 12.
Actual use in human
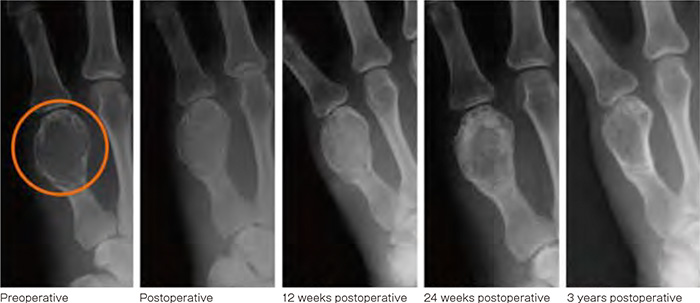
The area of the bone tumor lesion in the left fifth toe (shown with a circle) was surgically replaced with the new artificial bone. As a result, the artificial bone integrated with the surrounding bone and reduced the bone deformation caused by tumor.
Expectation for wide use in regenerative medicine and expansion of market
Because of reasons, such as the revision of Pharmaceutical Affairs Law during the clinic al experiments, 10 years were spent in developing this material as a product. This artificial bone gained market approval for medical devices in June 2012 under the proprietary name Refit and was covered by insurance since January 2013. Currently, the use of the product in surgery has commenced in some medical institutions.
This artificial bone that had overcome the problems associated with conventional products is not only expected to substitute for the products but also for use in a wide range of areas including regenerative medicine. The scale of Japanese market for artificial bones used in bone transplant treatment is said to be 10 billion yen, however the more widespread use of this new artificial bone is expected to increase the percentage of use, thereby accelerating the growth of the market.
