Research Results
’Self-eating’ cells: An intracellular bulk degradation process
Fascinated by AutophagyFY2017

- Noboru Mizushima (Professor, Graduate School and Faculty of Medicine, The University of Tokyo)
- PRESTO
- Unit Process and Combined Circuit “Molecular mechanism of autophagy and its physiological role” Researcher (1999-2002)
- PRESTO
- Time’s Arrow and Biosignaling “The Role of Autophagy in Mammals and Its Control Mechanism” Researcher (2002-2005)
- SORST
- “Intercellular Clearance Mechanism by Autophagy” Principal Researcher (2006-2007)
- ERATO
- “Mizushima Intracellular Degradation” Research Director (2017-2022)
Autophagy research, which was limited in research methods
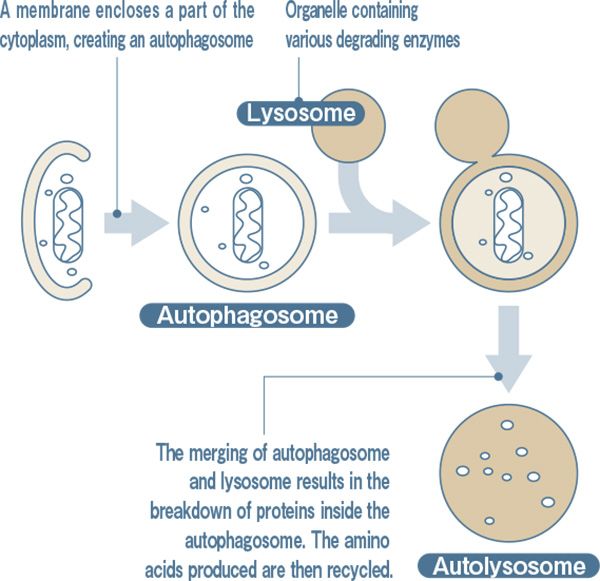
A cell eats itself. It was in the 1960s that this strange phenomenon was first discovered. Using an electron microscope, a cell eating the protein that was contained in the cell itself, and then breaking it down into amino acid was confirmed. A membrane encloses a part of the cytoplasm (autophagosome), which then becomes merged (autolysosome) with an organelle (lysosome) containing various degrading enzymes, causing the contained proteins to be broken down and becoming amino acids.
This phenomenon, named autophagy, was one that differed in nature compared to prior physiological knowledge. Tens of thousands of different protein types exist within our bodies, and these substances carry out various key functions. We eat every day, and the protein contained in these foods are broken down into as amino acids and absorbed. Then, the absorbed amino acids become components of protein synthesis, which is essential for life sustaining activities. Considering this common knowledge, it may seem like an unwise action of eating itself. There was speculation that this action involved metabolic action of transforming unnecessary protein into a different substance, but back then the only method available to observe this was through an electron microscope. It was difficult to confirm when, where and how this was taking place. As the years went by, autophagy would rarely be brought up by biologists researchers, and by the mid 90’s it had come to the point where only a few papers were published annually.
Autophagy
Development of a real-time observation technique using an animal cell
It was Professor Noboru Mizushima, who took an interest in autophagy at a time when few researchers addressed this subject. At that time he was still a graduate student at the Tokyo Medical and Dental University. It started out when he learned of the research by Professor Yoshinori Ohsumi of the National Institute for Basic Biology in Okazaki City (currently a Honorary Professor at the Tokyo Institute of Technology). Having confirmed the occurrence of autophagy in budding yeast, Professor Ohsumi had been carrying out his research. Professor Mizushima was drawn to this phenomenon called autophagy after coming across Professor Ohsumi’s paper, and he became a part-time researcher at Ohsumi’ s laboratory. This is how Professor Mizushima began his research.
One year later, he was selected to the PRESTO ‘Unit Process and Combined Circuit’ project, and he accelerated his research. He unraveled the mechanism of two proteins called the Apg12 and Apg5 bonding together to form an autophagosome. Even more ground-breaking was that he confirmed the occurrence of autophagy in tissues throughout the body even in mammals, which he accomplished with a technique for visualization in cells that using GFP. Prior to this, autophagosomes could only be captured with an electron microscope due to its small size. It had become possible to observe when, where and how autophagy is occurring in real-time. In the PRESTO ‘Time's Arrow and Biosignaling’ project to which he was selected thereafter, he engaged in investigating the roles of autophagy and its control mechanism within mammals by creating a knockout mouse that deficient in genes involved in autophagy.
Clarification of its physiological roles in maintaining homeostasis and starvation state
With the real-time observation and development of the knockout mouse, autophagy research progressed rapidly. Based on the observations that autophagy becomes active in the entire body when starved, and that a knockout mouse deficient in Atg5, a gene critical required in autophagy, falls into a severely undernourished condition immediately after birth and dies, it was found that autophagy plays an important alimentary role in starvation state. Also, a movement disorder was seen in mice that were deficient in Atg5 genes specific to nerve cells. Based on the abnormal accumulation and aggregation of protein seen when observing the inside of the nerve cell, it was found that in addition to supplementing amino acid during a starvation state, autophagy plays a housekeeping role intracellularly in removing misfolded or aggregated unnecessary proteins.
After the completion of the PRESTO project, Professor Mizushima continued his research at the SORST program entitled ‘Intracellular Clearance Mechanism by Autophagy’. Where he focused on autophagy's role in removing unnecessary proteins, and studied their roles in various tissues. For instance, in the fertilized eggs of mice, it was confirmed that autophagy becomes unusually active within four hours from fertilization. As for mouse deficient in Atg5 genes specific to egg cells, ovulation and fertilization occurred normally, but development stopped prior to reaching implantation and died as a result. In a normal mouse, the activation of autophagy immediately following fertilization allows the removal of unnecessary proteins that comes from the mother, as well as the securing of amino acids that are to become components of the new protein synthesis, prerequisite for the development process.
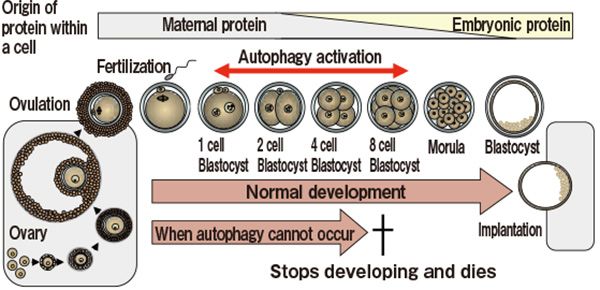
In the fertilized egg of a mouse in which autophagy does not occur, the development terminates before implantation and results in death
Clarifying the molecular mechanism of autophagy
Analysis of the molecular mechanism of autophagy in vertebrates is progressing. In the “MIZUSHIMA Intracellular Degradation” ERATO project, a novel autophagy factor that does not exist in yeast was discovered through a human genome-wide comprehensive screen by using genome editing technology. Furthermore, an autophagy receptor that selectively recognizes the endoplasmic reticulum was also discovered. The project is conducting cross-disciplinary research from a new perspective based on multiple methods including physics and molecular evolution.
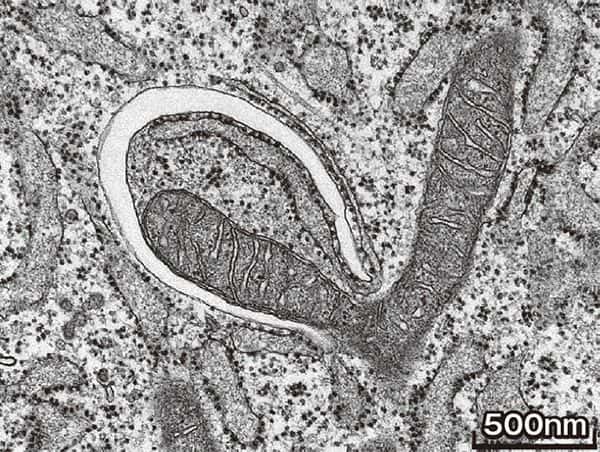
The moment an autophagosome is formed, as captured by an electron microscope. The mitochondria (V-shape in the center), is about to be enclosed by the white - appearing isolation film (upper left part of center). (Photo: Chieko Kishi)
An emerging research field that is attracting worldwide attention
In this way, Professor Mizushima and his colleagues used a newly developed observation method to reveal that autophagy is evolutionarily conserved in eukaryotesfrom yeast to mammals, and they have continued to unravel its mechanism and role. This contributed to the promotion of autophagy research all over the world, and is about to lead us to treatment for diseases such as cancer and Alzheimer’s. In 2013, Professor Mizushima was jointly awarded with Honorary Professor Ohsumi and others as Highly Cited Researchers selected by Thomson Reuters. The selection criteria for this award is based on the number of times a researcher's own paper was cited in others papers, and honorands are said to be leading candidates to receive a Nobel prize. Several decades have passed since the time when only a few related papers would be published each year. With the research results of Professor Mizushima and others who possess foresight as the impetus, autophagy is growing into a research area attracting the attention of the world.
- Life Science
- Research Results
- Japanese
