PRESTO PhaseI
Munehiko Asayama

- Professor, School of Agriculture, Ibaraki University
Basic studies for overproduction and recovery of value-added materials from autolysed cyanobacterial cells, cultivated under exhaust CO2-gas condition
A unique cyanobacterium ABRG5-3, filamentous and nonheterocystous srain, has been isolated. This strain allows transconjugation with expression vectors and showed an abundant accumulation of pigments, easy recovery of nucleic acids, and autolysed cells under static cultivation. Since autolysis can be quite convenient for easy recovery of overproduced materials from the cells without complicated cell-destruction and centrifugation, we try to obtain transconjugants of ABRG5-3 overproducing value-added materials and to recover the products from the autolysed cells. A new process involving cyanobacterial biotechnology may contribute to the recycled use of carbon resources and advances in industry.

Hiroki Ashida

- Associate Professor, Graduate School of Human Development and Environment Kobe University
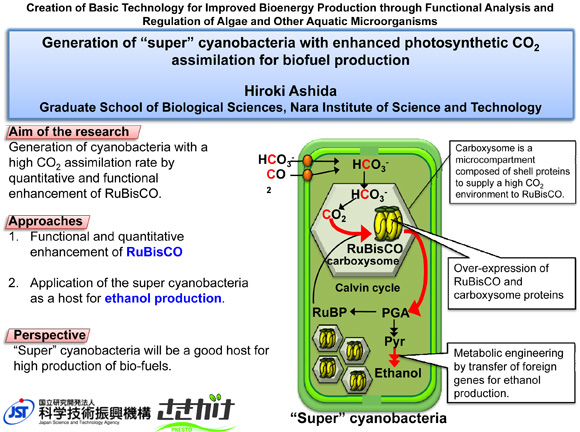
Generation of "super" cyanobacteria with enhanced photosynthetic CO2 assimilation for biofuel production
Cyanobacteria have recently gained great attention as promising biofuel-producing organisms. In order to utilize cyanobacteria for a sustainable energy production future, the improvement of their productivity through enhancement of photosynthesis is required. This research aims to create "super" cyanobacteria with increased photosynthetic carbon assimilation rate by quantitative enhancement of the CO2-fixing enzyme, RuBisCO and the carboxysome which provides a high CO2 environment for RuBisCO packaged inside of this microcompartment. Super cyanobacteria obtained here are applied for ethanol production.
Yutaka Amao

- Professor, Advanced Research Institute for Natural Science and Technology, Osaka City University
Development of photosynthesis organ from Algae assembled electrode device for biofuel conversion system
Photovoltaic conversion system with the function of carbon dioxide reduction using photosynthesis organ or Thylakoid membrane obtained from green algae Spirulina, which has effective oxygenetic photosynthesis function, assembled onto electrode will be demonstrated in carbon dioxide saturated water as an electrolyte. Moreover, innovative photon-to-current conversion system with the function of low carbon fuel methanol production will be developed using photosynthesis organ or Thylakoid membrane assembled onto electrode.

Takashi Osanai

- Senior Assistant Professor, Department of Agricultural Science, School of Agriculture, Meiji University
Increased production of cyanobacterial PHB by modification of dynamics of sugar metabolism
Bioplastics, which is made from biomass, is indispensable for overcoming global enviromental problem. In this study, I try to increase the productivity of bioplastic PHB using unicellular cyanobacteria.In order to change metabolic flow,I will generate cyanobacterial strains with modified transcriptional reuglators, resulting in production of safe and cheap bioplastics.

Hideki Kanda

- Assistant Professor, Department of Chemical Engineering, Nagoya University
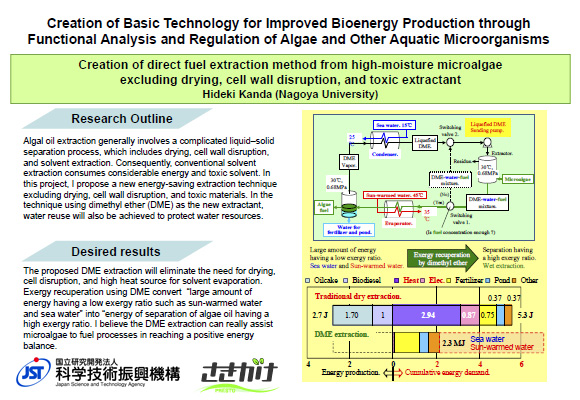
Creation of algae fuel direct extraction method from high-moisture microalgae without drying, cell wall disruption, and toxic extractant
Microalgae have a high capacity for absorbing carbon dioxide. However, a huge amount of water remains in microalgae, even after mechanical dewatering processes. The existing algae fuel production process include drying, cell wall disruption, and toxic extractant. These steps are energy-consuming and environmentally damaging. In this project, an eco-friendly algae fuel extraction technology without drying, cell wall disruption, and toxic extractant will be created by using dimethyl ether as an extractant. In this method, recycling of water will also be acheived to protect water resources.
Tatsuya Tomo

- Professor, Faculty of Science, Tokyo University of Science
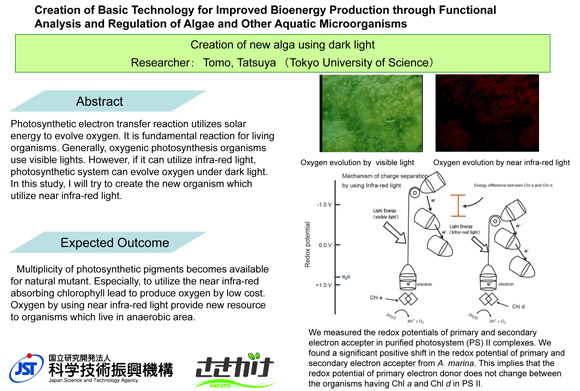
Creation of new alga using dark light
Photosynthetic electron transfer reaction utilizes solar energy to evolve oxygen. It is fundamental reaction for living organisms. Generally, oxygenic photosynthesis organisms use visible lights. However, if it can utilize infra-red light, photosynthetic system can evolve oxygen under dark light. In this study, I will try to create the new organism which utilize infra-red light.
Yuki Nakamura

- Assistant Research Fellow, Institute of Plant and Microbial Biology, Academia Sinica
Establishment of novel technical platforms on the triglyceride metabolic engineering in eukaryotic algae.
This project aims to build up novel technical platforms on the algal triglyceride engineering for biodiesel production. Basic studies on molecular mechanisms underlying the biosynthesis and accumulation of the triglycerides will be followed by the establishment of an innovative technical basis to mass-produce triglycerides with given quality.

Tomohisa Hasunuma

- Professor, Organization of Advanced Science and Technology, Kobe University
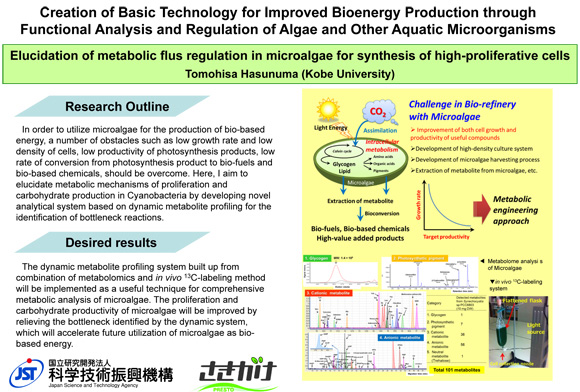
Elucidation of metabolic flux reguration in microalgae for synthesis of high-proliferative cells
Microalgae are able not only to produce energy from CO2 using sun light energy but also to avoid competition with fool supply and land utilization, which would profide a useful system for energy production from biomass. In the PRESTO study, by the development of a novel metabolic analysis technique applicable for the identification of rate limiting steps in metabolic pathways of microalgae, the identification of proliferative determinants is aimed to increase bio-energy supply from microalgae.
Yukako Hihara

- Associate Professor, Graduate School of Science and Engineering, Saitama University
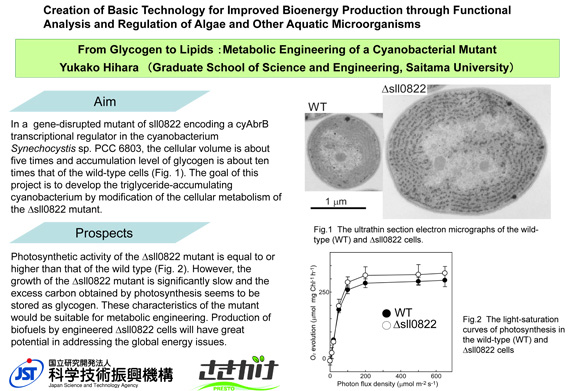
From glycogen to lipids: Metabolic engineering of a cyanobacterial mutant
The sll0822 gene in the cyanobacterium Synechocystis sp. PCC 6803 plays an important role in carbon/nitrogen balancing, and the cells of its disruption mutant are very large and accumulating ten times the level of glycogen in comparison with wild type. The goal of the project is to modify the cellular metabolism of the mutant to develop the triglyceride-accumulating cyanobacterium by the conversion of glycogen to triglyceride through fatty acid.
Kohsuke Honda

- Associate Professor, Department of Biotechnology, Graduate School of Engineering, Osaka University
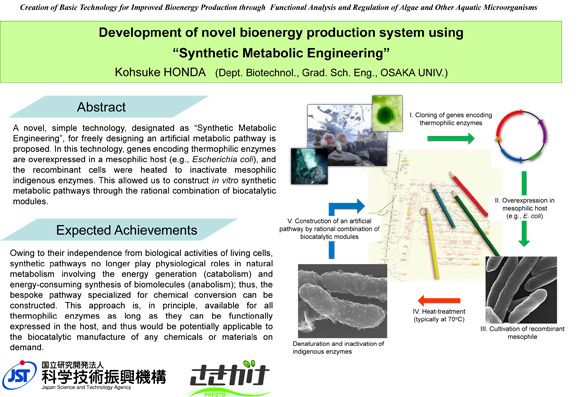
Development of novel bioenergy production system using "synthetic metabolic engineering"
This work aims to develop "synthetic metabolic engineering" strategies for production of value-added chemicals. Artificial biosynthetic pathways are constructed by the combined use of thermophilic metabolic enzymes, which are hetelorogously produced in mesophilic host strains (e.g. E. coli). By using them at high temperature (70 degree C or higher), in vitro fermentation without any byproducts can be achieved. This work particularly focuses on biobutanol production using this approach.
Hajime Masukawa

- Special-appointment Associate Professor, The OCU Advanced Research Institute for Natural Science and Technology (OCARINA), Osaka City University
Genetic engineering of cyanobacteria to enhance photobiological production of hydrogen
Cyanobacteria use sunlight and water to produce hydrogen gas (H2), which is potentially useful as a clean and renewable biofuel. Photobiological H2 arises primarily as an inevitable by-product of N2 fixation by nitrogenase, an oxygen-labile enzyme. In some filamentous cyanobacteria, the enzyme is localized to the microaerobic environment of heterocysts, a highly differentiated subset of the filamentous cells. Despite these advantages, the application of photosynthetic organisms to H2 production on an industrial scale has many challenges. The goal of this study is to enhance cyanobacterial production of H2 by genetic manipulation of nitrogenase and increase of heterocyst frequency.


